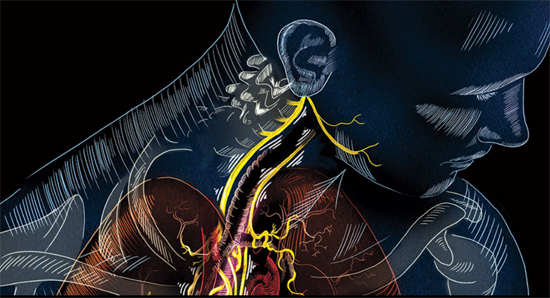
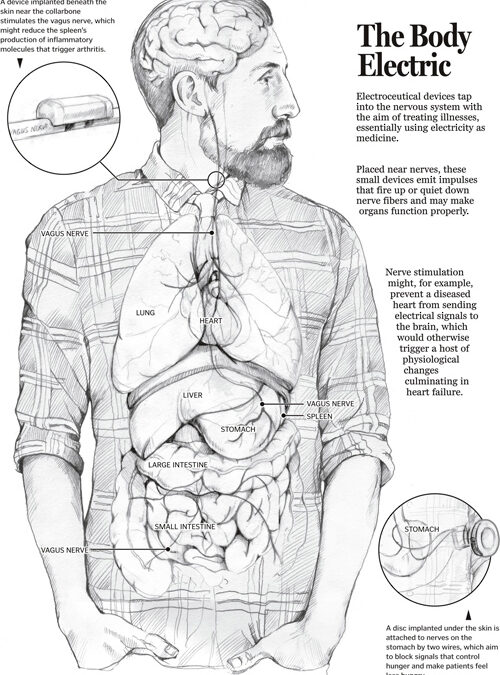
PART 1 PART 2 The CES ultra underscores its use of its conductive rubber ear clips. The rationale behind it is some interesting science on the ear; especially the vagus nerve and the special role it plays in the body. Read on. Ref: Wandering nerve could lead to range...